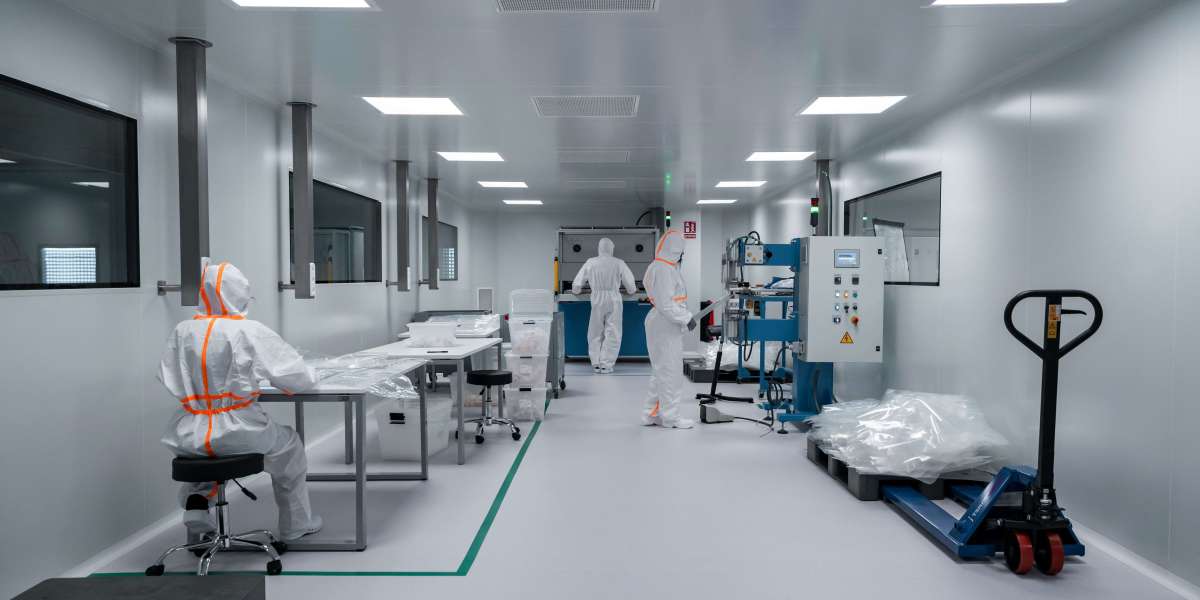
In the rapidly evolving world of high-tech manufacturing, the difference between a successful product launch and a costly recall often comes down to what you cannot see. From the assembly of life-saving medical devices to the precision of nanotechnology, the air we breathe is filled with invisible antagonists—dust, airborne organisms, and vaporized particles—that can compromise product integrity. To combat these threats, industries rely on strictly controlled environments, yet navigating the regulatory landscape for iso cleanrooms can be a complex challenge for many manufacturers.
Whether you are operating in Canada or the United States, understanding the intersection of international standards and local microbial limits is essential for operational success and regulatory compliance. This guide explores the technical requirements, classification systems, and engineering solutions necessary to maintain world-class sterile environments.
The Global Blueprint: Understanding ISO 14644
The foundation of modern contamination control is the ISO 14644 standard, which covers cleanrooms and associated controlled environments. This international framework provides a rigorous classification system consisting of nine distinct levels. Each level represents a magnitude of difference in allowable particle concentrations, providing a common language for engineers and regulators worldwide.
However, ISO standards serve as a baseline rather than a one-size-fits-all solution. What qualifies as a "clean room" in one sector may be considered insufficient in another, depending on the jurisdiction and the specific product being manufactured. In North America, manufacturers must layer local "Good Manufacturing Practices" (GMP) on top of these ISO classifications to meet the safety expectations of health authorities.
Canadian Microbial Limits: The Health Canada Framework
In Canada, requirements for sterile environments are determined by Health Canada through the Food and Drug Act and its embedded regulations. Specifically, the Food and Drug Regulations (Division 2) define "Sterile Products" as those requiring manufacturing in separate, enclosed areas under the supervision of personnel trained in microbiology.
While the core regulations focus on the definition of sterility, the technical roadmap is found in the Good Manufacturing Practices Guide for Drug Products (GUI-0001). Here, Health Canada defines four specific grades of clean rooms, which differ from the nine ISO levels but utilize ISO methods for sampling and demonstrating conditions.
The Four Grades of Cleanliness
The Canadian system categorizes cleanliness into four grades, often mapped to their ISO equivalents:
- Grade A: The most critical zone for high-risk operations, limited to 3,520 particles (≥0.5μm) per cubic meter.
- Grade B: Typically used for aseptic preparation and filling; this is the equivalent of an ISO 5 environment.
- Grade C: A standard for less critical stages of sterile manufacturing, equivalent to ISO 7.
- Grade D: The least restrictive sterile grade, equivalent to ISO 8.
One of the most important nuances of the Canadian approach is the distinction between "at rest" and "operational" states. Health Canada recognizes that a facility’s microbial profile changes when machinery is running and personnel are present, requiring distinct limits to ensure safety during the actual manufacturing process.
Bridging the Gap: American Regulations and the FDA
Manufacturers familiar with the Canadian system will find many similarities in the United States, though the regulatory bodies differ. In the U.S., the Food and Drug Administration (FDA) oversees clean room requirements through the Current Good Manufacturing Practices (CGMP).
Specifically, Code of Federal Regulations (CFR) 210 and 211 provide the "overhead" requirements for the production of sterile products. These regulations focus on the equipment used to control air pressure, micro-organisms, dust, and humidity, as well as the implementation of advanced air filtration systems. While Canada strictly utilizes a 0.5μm particle size threshold, some industries in the United States may use a 1.0μm threshold, making it vital for cross-border manufacturers to tailor their iso cleanrooms to the most stringent requirements of their target market.
Technical Specs in Action: The ISO Class 7 Standard
To understand the practical application of these standards, we can look at the recent expansion of Class 7 facilities in the medical device sector. An ISO Class 7 clean room (equivalent to Health Canada's Grade C) is a specialized environment designed to maintain extremely low levels of particulates.
For a facility to achieve and maintain Class 7 status, the engineering must meet several critical benchmarks:
- Particle Limits: Air cleanliness must be maintained at a maximum of 10,000 particles (≥0.5µm) per cubic foot.
- Filter Coverage: HEPA filtration systems must provide between 15% and 25% filter coverage of the ceiling area.
- Air Changes: The system must deliver a minimum of 60 air changes per hour.
- Air Flow Velocity: The standard air flow rate should range between nine and 16 cubic feet per minute (CFM) per square foot.
These rigorous standards allow companies like Sterling Industries to perform high-complexity activities such as Tyvek-sealed packaging, ultrasonic welding, UV bonding, and complex medical device assembly.
Engineering the Solution: Advanced Air Purification
The cornerstone of meeting any microbial limit is the air purification system. Solutions like the Air-Fit by Aeroex utilize ceiling-mounted fan filtration units equipped with centrifugal fans and HEPA filters to meet all modern clean room standards.
High-efficiency filtration is critical because it often exceeds the baseline regulatory requirements. For example, while Canadian standards focus on 0.5μm particles, high-quality HEPA filters offer a 99.99% efficiency rating for particles as small as 0.3μm. This provides a "safety buffer" for manufacturers aiming for the highest levels of sterility.
For larger facilities, these units can be deployed in a series with capacities ranging from 500 to 1,000 CFM. Modern systems also include central control panels, allowing operators to monitor and configure multiple units simultaneously. This is particularly useful for managing the "at rest" versus "operational" modes required by Health Canada, ensuring the facility remains compliant while optimizing energy use.
Conclusion
In the modern industrial landscape, maintaining strict microbial limits is more than a regulatory hurdle; it is a fundamental pillar of product quality and consumer safety. By understanding the intricate layers of ISO 14644, Health Canada’s grading system, and the FDA’s CGMP requirements, manufacturers can build facilities that are both compliant and highly efficient.
As technologies continue to advance in fields like medical devices and nanotechnology, the demand for ultra-pure environments will only grow. Investing in robust iso cleanrooms and high-efficiency HEPA filtration ensures that your facility is prepared to meet the challenges of today and the even stricter standards of tomorrow