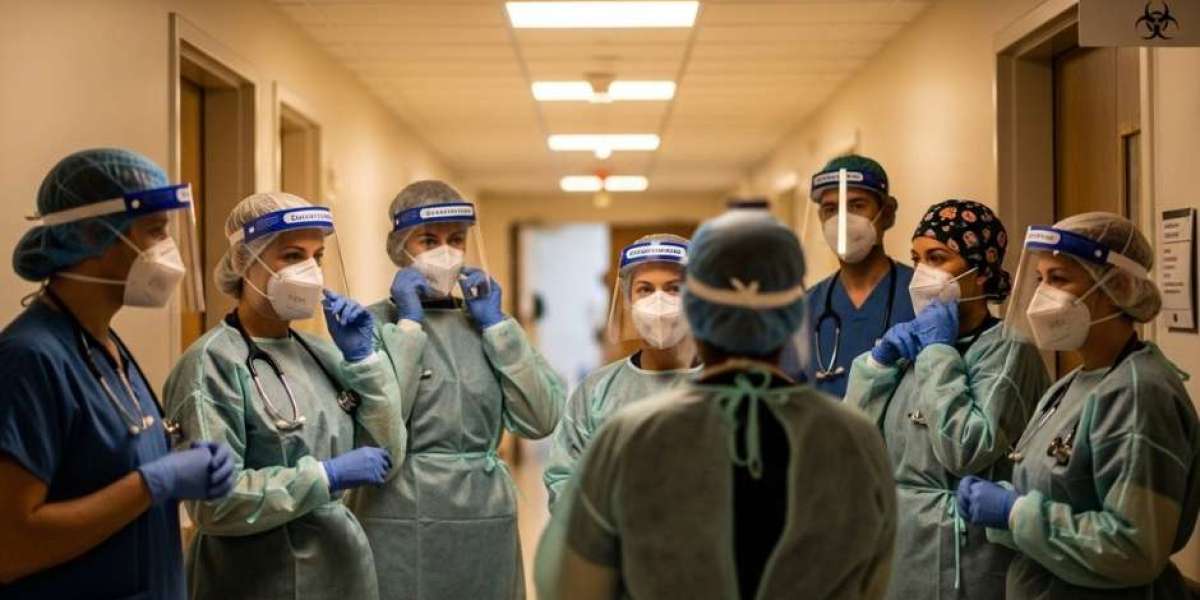
In any facility where patients are treated, samples are handled, or infectious materials are present, protective garments are the first and most visible line of defense against the spread of infection. Among the most critical of these garments is the isolation gown — a single-use or reusable protective garment worn by healthcare workers, lab technicians, and support staff to prevent the transfer of microorganisms, bodily fluids, and contaminants between patients, staff, and the wider environment.
As healthcare facilities face mounting pressure to strengthen infection prevention protocols, reduce operational overhead, and meet increasingly stringent regulatory standards, disposable isolation gowns have emerged as the preferred choice for administrators and procurement teams worldwide. Whether you are evaluating a new disposable isolation gown manufacturer or reviewing your facility's current PPE program, this guide explains what disposable isolation gowns are, how they work, and why your facility needs them.
What Are Disposable Isolation Gowns?
Disposable isolation gowns are single-use protective garments designed to create a barrier between the wearer and potential sources of infection or contamination. They are worn over regular clothing or scrubs and cover the torso, arms, and wrists — the areas most likely to come into contact with infectious material during patient care or laboratory work.
Unlike reusable gowns that are laundered and recirculated, disposable isolation gowns are discarded after a single use. This eliminates the risk of contamination transfer between uses and ensures that every garment meets its original barrier specification when worn.
Disposable isolation gowns are manufactured from a range of non-woven materials, including polypropylene, SMS (spunbond-meltblown-spunbond), polyethylene-coated fabric, and microporous laminates. Each material offers a different combination of fluid resistance, breathability, and durability — allowing facilities to select the appropriate gown for specific clinical or laboratory tasks.
Understanding the Classification
Not all isolation gowns offer the same level of protection. In the United States and internationally, isolation gowns are classified according to the Association for the Advancement of Medical Instrumentation (AAMI) standard PB70, which defines four levels of barrier protection based on liquid resistance testing.
- Level 1 gowns provide minimal protection and are suitable for basic care tasks with low fluid exposure risk — such as standard medical ward rounds or visitor use.
- Level 2 gowns offer low-to-moderate protection and are appropriate for blood draws, pathology labs, and ICU care.
- Level 3 gowns provide moderate-to-high protection for arterial blood draws, emergency care, and trauma situations involving significant fluid exposure.
- Level 4 gowns offer the highest protection level and are used in surgical environments, high-risk infection control situations, and prolonged fluid exposure scenarios.
When sourcing from a disposable isolation gown manufacturer, confirming the AAMI protection level of each product is essential to matching the garment to the specific risk profile of each clinical or operational environment.
How to Wear a Surgical Gown: The Correct Protocol
Understanding how to wear surgical gown correctly is as important as selecting the right garment. Even the highest-rated isolation gown provides inadequate protection if donned or removed incorrectly. The following is the standard protocol followed in clinical and regulated environments.
Before donning the gown, perform hand hygiene thoroughly. Remove the gown from its sealed packaging carefully to avoid contact with the outer surface. Hold the gown at the shoulder seams and allow it to unfold fully without touching the floor or any unclean surfaces. Slide both arms into the sleeves simultaneously, ensuring the gown covers the torso completely from neck to mid-thigh or knee level.
Secure the neck tie or Velcro fastening at the back of the neck first, followed by the waist ties or back closure. Ensure the gown overlaps fully at the back with no exposed clothing or skin. If gloves are part of the PPE ensemble, pull the cuff of the gloves over the wrist cuff of the gown to create a continuous barrier from sleeve to fingertip.
Doffing — the removal of the gown — requires equal care. The outer surface of a used isolation gown is considered contaminated and must not touch the face, scrubs, or unprotected skin during removal. Untie the waist ties first, followed by the neck fastening. Roll the gown inward from the shoulders, keeping the contaminated outer surface folded inside, and dispose of it immediately in the appropriate clinical waste container. Perform hand hygiene immediately after disposal. Knowing how to wear surgical gown and remove it correctly is a fundamental competency for all clinical and laboratory staff — and facilities should incorporate this into regular PPE training programs.
Why Your Facility Needs Disposable Isolation Gowns
Infection Prevention at the Core
The primary function of an isolation gown is to prevent the transmission of infection — from patient to healthcare worker, from worker to patient, and from one patient environment to another. Disposable isolation gowns fulfill this function more reliably than reusable alternatives because their barrier properties are guaranteed at every use. There is no degradation from repeated laundering, no uncertainty about whether the decontamination cycle was adequate, and no risk of residual contamination from a previous use. In facilities managing immunocompromised patients, infectious disease units, or surgical environments, this reliability is non-negotiable.
Regulatory Compliance & Accreditation
Healthcare facilities operating under Joint Commission accreditation, CMS conditions of participation, or national infection control standards are required to maintain documented PPE programs that demonstrate appropriate barrier precautions. Sourcing from a certified disposable isolation gown manufacturer ensures that your garments come with the conformity declarations, AAMI level certifications, and FDA 510(k) clearances required for regulatory compliance. This documentation simplifies audit preparation and removes the compliance uncertainty that comes with reusable garment programs.
Operational Efficiency & Cost Control
The operational case for disposable isolation gowns is as strong as the clinical one. Reusable gowns require laundering, inspection, repair, and redistribution — a resource-intensive cycle that consumes staff time, laundry capacity, and budget. Disposable gowns replace this entire process with a straightforward dispense-and-dispose model. Bulk procurement from a reliable disposable isolation gown manufacturer delivers competitive per-unit pricing, consistent supply, and predictable inventory management. For facilities managing high patient volumes or surge capacity scenarios, the ability to rapidly scale gown supply without laundering constraints is a critical operational advantage.
Readiness for Outbreak & Surge Scenarios
The COVID-19 pandemic exposed the vulnerability of healthcare supply chains built around reusable PPE. Facilities that had established relationships with disposable isolation gown manufacturers and maintained strategic stock of single-use gowns were significantly better positioned to protect staff and patients during surge periods. Building disposable isolation gowns into your standard PPE inventory — alongside your broader surgical disposable items list — ensures your facility is prepared for unexpected demand spikes without compromising protection standards.
Staff Confidence & Patient Trust
Healthcare workers who are properly equipped with clean, correctly specified protective garments perform with greater confidence. Patients, too, are reassured by visible evidence that infection control protocols are being rigorously followed. Disposable isolation gowns — fresh, uniform, and visibly clean — communicate a standard of care that worn or improperly laundered reusable garments simply cannot match.
Choosing the Right Disposable Isolation Gown Manufacturer
When evaluating a disposable isolation gown manufacturer for your facility, prioritize suppliers who offer full AAMI level certification across their product range, FDA registration and relevant regulatory clearances, transparent third-party testing documentation, consistent bulk supply capability with reliable lead times, and a responsive account team that can support your compliance and procurement needs.
The right manufacturer is not simply a product vendor — they are a clinical safety partner whose garments protect your staff and patients every single day.
Final Thoughts
Disposable isolation gowns are not a commodity purchase — they are a clinical safety decision. Understanding what they are, how they are classified, how to wear surgical gown correctly, and why single-use alternatives outperform reusable options across risk, cost, and compliance dimensions is essential for any healthcare administrator or procurement professional serious about infection prevention.
Review your current gown program against AAMI protection level requirements. Audit your supplier's compliance credentials. Ensure your staff are trained in correct donning and doffing procedures. And make disposable isolation gowns a core, non-negotiable element of your facility's infection control infrastructure.
Your staff deserve consistent protection. Your patients deserve consistent safety. Disposable isolation gowns deliver both.